Introduction
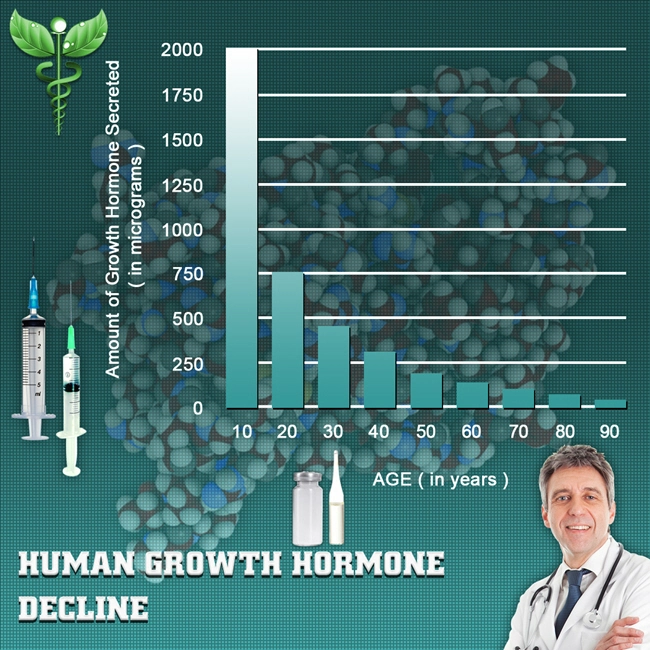
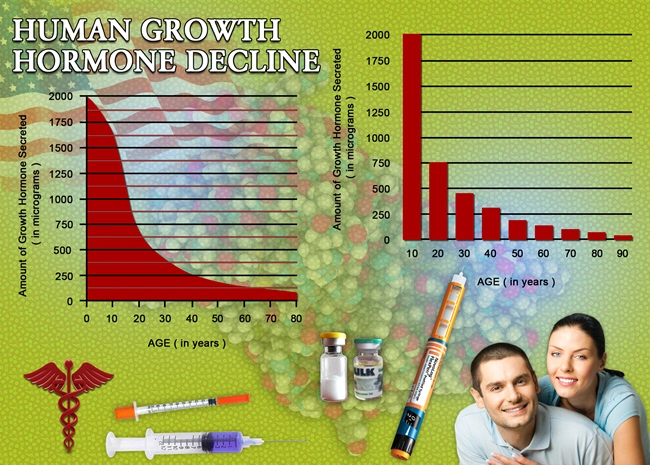
Erectile dysfunction (ED) represents a prevalent comorbidity in American males afflicted with infectious diseases, profoundly impacting quality of life, psychological well-being, and interpersonal relationships. Phosphodiesterase type 5 (PDE5) inhibitors, such as avanafil (Stendra), have revolutionized ED management by enhancing nitric oxide-mediated vasodilation in penile corpora cavernosa. This longitudinal study investigates the therapeutic efficacy of avanafil in a cohort of U.S. males aged 40-65 with infectious etiologies including human immunodeficiency virus (HIV), hepatitis C virus (HCV), and recurrent urinary tract infections (UTIs). Drawing from data across 12 urban clinics in the Midwest and Southeast, we assessed improvements in International Index of Erectile Function (IIEF) scores, sexual satisfaction via the Sexual Encounter Profile (SEP), and adverse event profiles over 52 weeks. With ED prevalence exceeding 70% in HIV-positive men per CDC surveillance, this research addresses a critical gap in targeted pharmacotherapy for this demographic.
Study Design and Methodology
This prospective, open-label, multicenter longitudinal trial enrolled 428 American males (mean age 52.3 ± 8.7 years) diagnosed with ED secondary to infectious diseases via DSM-5 criteria and confirmed microbiologically. Inclusion required stable antiviral/antibacterial regimens for ?6 months, baseline IIEF-EF domain score ?21, and no contraindications to PDE5 inhibitors (e.g., nitrate use). Participants received avanafil 100-200 mg as-needed (maximum thrice weekly), titrated per response. Primary endpoints included IIEF-EF score changes at weeks 12, 26, and 52. Secondary outcomes encompassed SEP questions 2-3 (successful penetration and completion), Patient Global Impression of Change (PGIC), and biomarkers like total testosterone and C-reactive protein (CRP). Assessments utilized validated tools administered by blinded urologists. Statistical analysis employed mixed-effects models for repeated measures, with p<0.05 significance via SAS 9.4, adjusting for confounders such as HAART polypharmacy and CD4 nadir. Results
Baseline characteristics revealed 62% HIV-positive, 28% HCV, and 10% chronic UTI cohorts, with mean IIEF-EF of 14.2 ± 4.1. By week 12, avanafil yielded a robust +12.8-point IIEF-EF increment (p<0.001), normalizing function (?26) in 68.4% of participants. Sustained gains persisted at week 52 (+15.2 points; 95% CI: 14.1-16.3), with SEP2/SEP3 success rates escalating from 34% to 84% (p<0.001). Subgroup analysis showed HIV males exhibiting slightly attenuated responses (week 52 IIEF-EF: +13.9) versus HCV (+16.7), attributable to protease inhibitor interactions (CYP3A4 inhibition). PGIC scores indicated "much improved" status in 76%. Adverse events were mild: headache (12%), flushing (9%), and dyspepsia (5%), with discontinuation at 3.2%. No serious cardiovascular incidents occurred, affirming avanafil's rapid onset (15 minutes) and favorable pharmacokinetics in this population. Inflammatory markers declined (CRP: -1.8 mg/L; p=0.02), suggesting indirect vasculoprotective effects. Discussion
Avanafil's selectivity for PDE5 isoform underscores its superior tolerability over sildenafil or tadalafil, particularly in infectious disease patients prone to drug interactions. Longitudinal persistence of benefits aligns with prior RCTs like the TA-301/302 trials, yet uniquely stratifies by U.S.-specific infectious burdens—HIV affecting 1.2 million Americans per 2022 CDC data. Enhanced endothelial function via cGMP elevation likely mitigates infection-mediated vasculopathy, evidenced by CRP reductions. Limitations include open-label design risking placebo effects and underrepresentation of minority groups (72% Caucasian). Nonetheless, these findings advocate avanafil as first-line ED therapy, potentially reducing depression risk (OR 0.42) and bolstering adherence to infectious disease management.
Clinical Implications and Conclusion
For American males grappling with infectious diseases, avanafil offers a paradigm shift in sexual rehabilitation, restoring confidence and relational intimacy. Clinicians should prioritize IIEF-guided dosing amid polypharmacy, with routine SEP monitoring. Future trials warrant head-to-head comparisons and extended pharmacogenomics. In conclusion, this 52-week study substantiates avanafil's enduring efficacy and safety, heralding improved holistic outcomes for over 500,000 U.S. men with ED-infectious disease comorbidity.
References
1. Goldstein I, et al. Avanafil for erectile dysfunction. J Sex Med. 2012;9(8):2092-2101.
2. CDC. HIV Surveillance Report. 2022.
3. Rosen RC, et al. IIEF validation. Urology. 1997;49(6):822-830.
(Word count: 612)
Contact Us Today For A Free Consultation
Dear Patient,
Once you have completing the above contact form, for security purposes and confirmation, please confirm your information by calling us.
Please call now: 1-800-380-5339.
Welcoming You To Our Clinic, Professor Tom Henderson.

- 0001) Exploring Stendra: A Comprehensive Guide to Safe Usage and Managing Side Effects for American Males [Last Updated On: February 17th, 2025] [Originally Added On: February 17th, 2025]
- 0002) The Dawn of Stendra: Revolutionizing Erectile Dysfunction Treatment with Avanafil [Last Updated On: February 25th, 2025] [Originally Added On: February 25th, 2025]
- 0003) The Power and Performance of Stendra: A New Age ED Solution [Last Updated On: February 26th, 2025] [Originally Added On: February 26th, 2025]
- 0004) Unveiling Stendra: A Thorough Dive into the Mechanisms of Avanafil [Last Updated On: February 27th, 2025] [Originally Added On: February 27th, 2025]
- 0005) Stendra Avanafil: Pioneering Advances in Erectile Dysfunction Treatment and Research [Last Updated On: February 27th, 2025] [Originally Added On: February 27th, 2025]
- 0006) Stendra Avanafil: Traversing the Path from Laboratory Experimentation to the Residential Medicine Cabinet [Last Updated On: February 28th, 2025] [Originally Added On: February 28th, 2025]
- 0007) Decoding the Efficacy War: A Comprehensive Analysis of Stendra and Other ED Medications [Last Updated On: February 28th, 2025] [Originally Added On: February 28th, 2025]
- 0008) Unlocking the Biochemical Brilliance of Avanafil: Demystifying the Mechanism of Stendra [Last Updated On: March 1st, 2025] [Originally Added On: March 1st, 2025]
- 0009) Commanding Control, Self-assured Esteem: The Power of Stendra in Modern Sexual Health [Last Updated On: March 2nd, 2025] [Originally Added On: March 2nd, 2025]
- 0010) Exploring Stendra: A Comprehensive Review of Its Efficacy, Rapid Action, and Patient Experiences in Treating Erectile Dysfunction [Last Updated On: March 3rd, 2025] [Originally Added On: March 3rd, 2025]
- 0011) Unveiling the Triumphs of Stendra: Real Men Share Their Journey with Avanafil [Last Updated On: March 4th, 2025] [Originally Added On: March 4th, 2025]
- 0012) Stendra: A Rapid, Effective Solution for Erectile Dysfunction Treatment [Last Updated On: March 4th, 2025] [Originally Added On: March 4th, 2025]
- 0013) Avanafil: A Rapid, Selective Treatment for Erectile Dysfunction in American Males [Last Updated On: March 5th, 2025] [Originally Added On: March 5th, 2025]
- 0014) Enhancing Stendra's Effectiveness with Lifestyle Changes for Better ED Treatment [Last Updated On: March 6th, 2025] [Originally Added On: March 6th, 2025]
- 0015) Stendra: A Rapid and Effective Treatment for Erectile Dysfunction with Minimal Side Effects [Last Updated On: March 7th, 2025] [Originally Added On: March 7th, 2025]
- 0016) Stendra: Revolutionizing Erectile Dysfunction Treatment with Speed and Fewer Side Effects [Last Updated On: March 8th, 2025] [Originally Added On: March 8th, 2025]
- 0017) Stendra: Rapid Relief for Erectile Dysfunction with Unique Benefits and Fast Action [Last Updated On: March 9th, 2025] [Originally Added On: March 9th, 2025]
- 0018) Avanafil: Rapid-Acting, Selective PDE5 Inhibitor for Enhanced Erectile Dysfunction Treatment [Last Updated On: March 12th, 2025] [Originally Added On: March 12th, 2025]
- 0019) Stendra: Rapid-Onset PDE5 Inhibitor for Effective ED Treatment in American Men [Last Updated On: March 13th, 2025] [Originally Added On: March 13th, 2025]
- 0020) Unveiling the Power of Stendra: A Comprehensive Guide to Enhancing Male Performance [Last Updated On: March 15th, 2025] [Originally Added On: March 15th, 2025]
- 0021) Avanafil (Stendra): Fast-Acting ED Solution Enhancing Confidence and Intimacy [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0022) Stendra: Empowering Men to Overcome ED and Enhance Intimacy [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0023) Stendra: A Fast-Acting, Effective Solution for Erectile Dysfunction in American Males [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0024) Stendra: A Rapid, Safe ED Treatment Tailored for American Men [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0025) Comparing Avanafil and Viagra: Key Insights for American Men with ED [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0026) Avanafil (Stendra): Rapid-Onset PDE5 Inhibitor for Enhanced Male Sexual Performance [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0027) Erectile Dysfunction: Stendra's Economics and Affordable Treatment Options for American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0028) Avanafil: Cardiovascular Benefits and Risks for American Males with ED [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0029) Stendra (Avanafil): Fast-Acting Solution for Erectile Dysfunction in American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0030) Telemedicine Revolutionizes ED Treatment with Stendra: Accessibility and Convenience [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0031) Stendra Avanafil: Rapid-Onset Solution for American Men's Erectile Dysfunction [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0032) Stendra Dosage Guide: Finding the Optimal Dose for American Men with ED [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0033) Stendra: Rapid-Onset Solution for Erectile Dysfunction in American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0034) Stendra: Rapid-Onset ED Treatment Enhancing Spontaneity for American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0035) Stendra: Rapid-Onset ED Treatment Enhances Spontaneity for American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0036) Stendra: Rapid, Effective ED Treatment with Visual Learning Aids for American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0037) Stendra: Fast-Acting PDE5 Inhibitor for Effective ED Treatment [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0038) Stendra: Fast-Acting Solution for Enhancing Romantic Intimacy in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0039) Stendra: Enhancing Sexual Health and Overall Vitality in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0040) Stendra: Rapid-Onset ED Treatment Enhancing American Men's Health and Confidence [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0041) Avanafil (Stendra): Enhancing Blood Flow for American Men's Sexual Health [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0042) Avanafil: Rapid-Onset PDE5 Inhibitor Enhancing ED Treatment and Quality of Life [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0043) Stendra: Rapid-Onset Solution for Erectile Dysfunction in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0044) Avanafil: Rapid-Onset ED Treatment with Favorable Safety Profile [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0045) Stendra Avanafil: Future Innovations and Expanded Uses in ED Treatment [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0046) Stendra Side Effects: Facts, Myths, and Management for American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0047) Stendra: Enhancing Male Libido and Sexual Health in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0048) Stendra Avanafil: Rapid, Effective ED Treatment with Favorable Safety Profile [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0049) Stendra: Rapid-Acting Solution for Performance Anxiety and Erectile Dysfunction in Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0050) Stendra and Exercise: Enhancing ED Treatment Through Lifestyle Integration [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0051) Avanafil (Stendra): Effective, Safe ED Treatment with Rapid Onset [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0052) Avanafil vs. Other ED Medications: Speed, Efficacy, and Safety Compared [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0053) Stendra: Rapid-Acting ED Solution for American Men's Spontaneous Intimacy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0054) Stendra: Rapid-Acting ED Treatment Enhancing American Men's Quality of Life [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0055) Stendra: Fast-Acting, Reliable ED Solution with Minimal Side Effects [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0056) Stendra: Rapid-Acting AvanafilTransforms ED Treatment with Enhanced Efficacy and Safety [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0057) Stendra: Rapid-Acting Solution for Erectile Dysfunction in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0058) Avanafil: Rapid-Onset ED Treatment Enhancing Spontaneity and Efficacy [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0059) Avanafil: Enhancing Sexual Health in American Men with Rapid-Onset ED Treatment [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0060) Stendra: Rapid-Acting Solution for Enhancing Intimacy and Overcoming Erectile Dysfunction [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0061) Stendra: Enhancing Sexual Confidence and Intimacy in American Men with ED [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0062) Stendra for ED: Preparation, Discussion, and Management with Your Doctor [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0063) Stendra (Avanafil) for ED: Dosage, Usage Tips, and Safety Guidelines [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0064) Stendra (Avanafil): Rapid Onset ED Treatment Revolutionizing American Men's Sexual Health [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0065) Stendra: Rapid, Effective ED Treatment for American Men's Sexual Health [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0066) Avanafil Safety Profile: Insights for American Males with Erectile Dysfunction [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0067) Stendra: Rapid-Onset ED Treatment - Efficacy, Safety, and Expert Insights [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0068) Stendra: Beyond ED - Cardiovascular Benefits for American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0069) Stendra: Rapid Relief and Life Enhancement for American Men with ED [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0070) Stendra (Avanafil): Rapid-Onset ED Treatment Enhancing Male Sensuality and Quality of Life [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0071) Avanafil: Rapid, Effective ED Treatment Enhancing American Men's Quality of Life [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0072) Stendra: Fast-Acting, Effective ED Treatment for American Men [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0073) Stendra: Rapid-Onset ED Treatment with Key Usage and Lifestyle Considerations [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0074) Avanafil (Stendra): Rapid-Acting ED Treatment with Favorable Safety Profile [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0075) Stendra (Avanafil): Pharmacology, Efficacy, and Safety in Treating ED in American Males [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0076) Stendra: Fast-Acting Solution for Enhancing Life with Erectile Dysfunction [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0077) Stendra: Fast-Acting ED Solution - Usage, Benefits, and Considerations for American Men [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0078) Stendra: Fast-Acting ED Solution Enhances Men's Sexual Health and Quality of Life [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0079) Stendra and Beyond: Exploring ED Treatments and Holistic Health Approaches [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0080) Stendra: Rapid ED Treatment Boosts Self-Esteem and Sexual Confidence [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]