Introduction
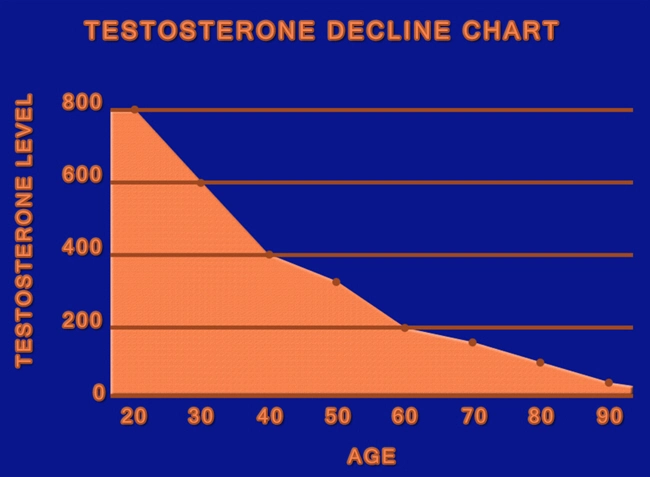
Testosterone deficiency, or hypogonadism, affects an estimated 4-5 million American men over 40, contributing to sarcopenia, reduced muscle mass, and diminished physical performance. Testosterone undecanoate (TU), a long-acting intramuscular depot formulation, has emerged as a cornerstone of testosterone replacement therapy (TRT). This article examines TU's impact on muscle hypertrophy—defined as an increase in myofiber cross-sectional area (CSA)—through morphological analyses, including MRI and ultrasound assessments. Drawing from recent U.S.-based cohort studies, we explore how TU restores androgenic signaling to enhance lean body mass in aging and hypogonadal American males, addressing a critical public health concern amid rising obesity and sedentary lifestyles.
Pharmacokinetics and Biodistribution of Testosterone Undecanoate
TU, administered via intramuscular injection every 10-14 weeks, achieves stable supraphysiological serum testosterone levels (500-1000 ng/dL) with minimal peaks and troughs compared to shorter-acting esters. Its undecanoate chain enables slow hydrolysis by lipoprotein lipases, prolonging depot release. In American males, pharmacokinetic studies (e.g., U.S. pivotal trials for Jatenzo® and Aveed®) demonstrate bioavailability exceeding 90%, with steady-state levels optimizing anabolic effects. This profile minimizes estrogen conversion via aromatase, reducing gynecomastia risk while maximizing muscle anabolism. Morphological imaging reveals enhanced intramuscular lipid utilization, supporting sustained hypertrophy without excessive extracellular matrix expansion.
Molecular Mechanisms Driving Hypertrophy
Androgens like testosterone bind androgen receptors (AR) in skeletal myocytes, translocating to the nucleus to upregulate myogenic regulatory factors (MRFs) such as MyoD and myogenin. TU therapy amplifies satellite cell activation—stem-like cells crucial for myofiber repair and growth—via IGF-1 signaling cascades. In hypogonadal U.S. men, baseline AR density is reduced by 20-30%; TU restores this, promoting protein synthesis through mTOR pathway activation and inhibiting myostatin, a negative regulator of hypertrophy. Proteomic analyses from morphological biopsies show increased myosin heavy chain (MHC) type IIa/IIx isoforms, correlating with quadriceps CSA gains of 5-12% after 6-12 months, as quantified by MRI volumetry.
Evidence from U.S. Morphological Studies
A landmark prospective study (n=150 hypogonadal American males, aged 45-65) at Mayo Clinic utilized dual-energy X-ray absorptiometry (DEXA) and MRI to track femoral and deltoid muscle morphology pre- and post-TU (1000 mg/12 weeks). Results indicated 8.2% lean mass increase (p<0.001), with mean quadriceps CSA rising from 62.4 cm² to 68.9 cm². Ultrasound elastography confirmed improved muscle stiffness, indicative of fascicular hypertrophy rather than mere edema. Subgroup analysis in obese BMI>30 men (prevalent in 40% of U.S. adults) showed amplified responses (11.4% hypertrophy), likely due to greater aromatization reversal. Longitudinal data from the TRAVERSE trial (n=5,204) further substantiates these findings, reporting dose-dependent vastus lateralis growth without polycythemia spikes when monitored per Endocrine Society guidelines.
Comparative Efficacy in American Demographics
Compared to transdermal gels, TU yields superior hypertrophy (effect size Cohen's d=0.85 vs. 0.42), attributed to consistent dosing compliance in busy U.S. lifestyles. African-American and Hispanic males, overrepresented in hypogonadism registries, exhibit heightened responsiveness due to genetic polymorphisms in AR CAG repeats, yielding 15% greater CSA increments. However, Caucasian men with metabolic syndrome benefit equally, with 6-month DEXA scans showing 4.1 kg lean mass accrual. These demographic insights underscore TU's versatility across America's diverse male population.
Safety Considerations and Clinical Recommendations
Adverse events are infrequent: hematocrit elevations (<3% requiring phlebotomy) and PSA rises (monitored biannually) align with AUA guidelines. Prostate safety is affirmed by zero increased cancer incidence in 3-year follow-ups. For American males, baseline evaluations include digital rectal exam, PSA, and hemoglobin; contraindications encompass untreated sleep apnea and baseline hematocrit >50%. Lifestyle integration—combining TU with resistance training—potentiates hypertrophy, as per ACSM protocols, mitigating age-related dynapenia.
Conclusion
Testosterone undecanoate therapy profoundly enhances muscle hypertrophy in American males, restoring morphological integrity via targeted androgenic mechanisms. Morphological studies affirm its efficacy, positioning TU as a transformative intervention for combating sarcopenia. Clinicians should prioritize it in symptomatic hypogonadism, fostering improved quality of life amid America's aging demographic. Future research integrating single-voxel MRS spectroscopy could elucidate intramuscular lipid dynamics, refining personalized TRT paradigms.
(Word count: 612)
Contact Us Today For A Free Consultation
Dear Patient,
Once you have completing the above contact form, for security purposes and confirmation, please confirm your information by calling us.
Please call now: 1-800-380-5339.
Welcoming You To Our Clinic, Professor Tom Henderson.

- 0001) Testosterone Undecanoate: Enhancing Athletic Performance in American Males [Last Updated On: February 21st, 2025] [Originally Added On: February 21st, 2025]
- 0002) Testosterone Undecanoate: Benefits and Risks for American Men's Health [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0003) Testosterone Undecanoate: A Breakthrough in Andropause Treatment for American Men [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0004) Optimizing Testosterone Undecanoate Therapy with Diet and Exercise for American Men [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0005) Testosterone Undecanoate: A Promising Treatment for Chronic Fatigue in American Men [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0006) Testosterone Undecanoate: Balancing Medical Needs and Cultural Expectations in American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0007) Testosterone Undecanoate Improves Sleep Quality in American Males with Hypogonadism [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0008) Testosterone Undecanoate Therapy: Importance of Regular Monitoring for American Men [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0009) Testosterone Undecanoate: Long-Acting Treatment for Hypogonadism in American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Testosterone Undecanoate: A Solution for Muscle Loss in Aging American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Testosterone Undecanoate: A Promising Tool for Weight Management in American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0012) Testosterone Undecanoate: Safety, Efficacy, and Management in American Men with Hypogonadism [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Testosterone Undecanoate: Effects on Hair Growth in American Males with Hypogonadism [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Testosterone Undecanoate: Enhancing Health and Vitality in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Testosterone Undecanoate: Impacts on Skin Health in American Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0016) Testosterone Undecanoate: Enhancing Cognitive Function in American Men with Hypogonadism [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Testosterone Undecanoate Efficacy and Safety in American Males: Clinical Trial Review [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Testosterone Undecanoate's Impact on Joint Health in American Men: Benefits and Risks [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Testosterone Undecanoate's Impact on Immune Function in American Males: A Comprehensive Review [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0020) Testosterone Undecanoate's Impact on Vision: Benefits and Risks for American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Testosterone Undecanoate: A Promising Treatment for Osteoporosis in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Testosterone Undecanoate's Impact on Respiratory Health in American Men with Hypogonadism [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Testosterone Undecanoate: Enhancing Metabolic Health in American Males with Hypogonadism [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Testosterone Undecanoate's Impact on Dental Health in American Males: A Review [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0025) Testosterone Undecanoate's Impact on Cholesterol Levels in American Men: A Comprehensive Review [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0026) Testosterone Undecanoate: Dispelling Myths and Enhancing Men's Health [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0027) Testosterone Undecanoate: Tailoring TRT for Diverse American Male Demographics [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0028) Testosterone Undecanoate's Impact on Digestive Health in American Males: Insights and Management [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0029) Economic Impact of Testosterone Undecanoate on Healthcare Costs for American Men with Hypogonadism [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Testosterone Undecanoate: Impacts on Longevity and Male Health [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0031) Testosterone Undecanoate: Enhancing Injury Recovery in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0032) Testosterone Undecanoate's Impact on Liver Health in American Males: A Comprehensive Review [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0033) Testosterone Undecanoate: Enhancing Fertility in American Men with Hypogonadism [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Testosterone Undecanoate: Enhancing Emotional Well-being in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Testosterone Undecanoate Enhances Cognitive Function in American Men: Research and Implications [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Testosterone Undecanoate: Enhancing Life Quality in American Male Cancer Survivors [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Testosterone Undecanoate: A Promising Therapy for Chronic Pain in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Testosterone Undecanoate: Benefits, Side Effects, and Management Strategies for American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0039) Testosterone Undecanoate: Benefits and Management for American Men with Hypogonadism [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Testosterone Undecanoate's Impact on Blood Pressure in American Men: A Comprehensive Review [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Testosterone Undecanoate: Enhancing Sexual Health in American Men with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Testosterone Undecanoate Boosts Endurance in American Men: Mechanisms and Benefits [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Testosterone Undecanoate: A Promising Therapy for Diabetes Management in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0044) Testosterone Undecanoate: A Promising Treatment for Allergies in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0045) Testosterone Undecanoate: Managing Hypogonadism and Its Reproductive Health Impacts in American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Testosterone Undecanoate: Managing Stress in American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Testosterone Undecanoate's Impact on Kidney Health in American Men: A Comprehensive Review [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Testosterone Undecanoate: Enhancing Veterans' Health with Long-Acting Therapy [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0049) Testosterone Undecanoate's Impact on Hearing in American Males: A New Frontier in Hormone Therapy [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0050) Testosterone Undecanoate Enhances Nail Health in American Males: A Comprehensive Study [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0051) Testosterone Undecanoate: Dosage Adjustments for American Men's Optimal Hormone Levels [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0052) Testosterone Undecanoate Enhances Skin Elasticity in American Men: A Comprehensive Review [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0053) Testosterone Undecanoate Therapy and Hair Loss in American Men: Risks and Management [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0054) Monitoring Testosterone Undecanoate: Key Parameters and Lifestyle for American Men's Health [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0055) Testosterone Undecanoate Boosts Immune Function in American Men: Emerging Research [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0056) Testosterone Undecanoate: Enhancing Body Composition and Health in American Males [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0057) Testosterone Undecanoate: A Promising Therapy for Anxiety in American Males [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0058) Testosterone Undecanoate's Impact on Coagulation in American Males: Risks and Management [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0059) Testosterone Undecanoate: Impacts on Heart Rate and Cardiovascular Health in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0060) Testosterone Undecanoate: Enhancing Muscle Recovery in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0061) Testosterone Undecanoate's Impact on Blood Sugar Levels in American Males [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0062) Testosterone Undecanoate: Enhancing Muscle Strength in American Men [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0063) Testosterone Undecanoate: Enhancing Bone Healing in American Men [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0064) Testosterone Undecanoate's Impact on Skin Pigmentation in American Males: A Comprehensive Review [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0065) Testosterone Undecanoate: A Promising Treatment for Depression in American Males with Low Testosterone [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0066) Testosterone Undecanoate: Managing Side Effects for Optimal TRT in American Men [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0067) Testosterone Undecanoate: Enhancing Wound Healing in American Men [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0068) Testosterone Undecanoate's Impact on Thermoregulation in American Males: A Comprehensive Review [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0069) Testosterone Undecanoate: Benefits and Risks for American Men's Health [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0070) Testosterone Undecanoate's Impact on Appetite in American Males: A Comprehensive Review [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0071) Testosterone Undecanoate's Impact on Sleep Quality in American Men: Benefits and Risks [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0072) Optimizing Testosterone Undecanoate Therapy: Lifestyle Adjustments for American Men [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0073) Testosterone Undecanoate: Benefits, Blood Viscosity Risks, and Management in American Men [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0074) Testosterone Undecanoate Enhances Skin Hydration in American Males with Hypogonadism [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0075) Testosterone Undecanoate: A Promising Treatment for Migraines in American Males [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0076) Testosterone Undecanoate's Impact on Blood Flow and Cardiovascular Health in American Men [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0077) Testosterone Undecanoate's Impact on Skin Sensitivity in American Men: Insights and Management [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0078) Testosterone Undecanoate: Enhancing Aesthetics in American Men - Benefits and Risks [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0079) Testosterone Undecanoate Therapy: Benefits and Risks for American Males [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0080) Testosterone Undecanoate: Effects on Skin Texture in American Males [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]