Introduction
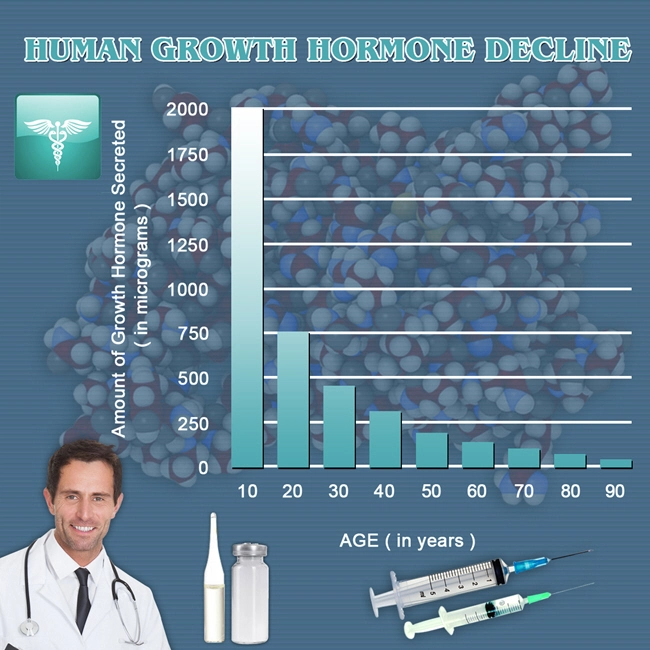
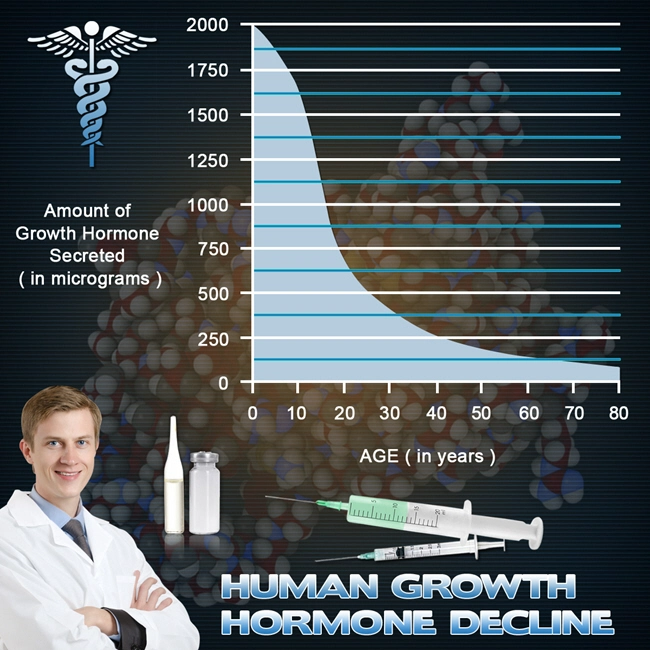
Coronary artery disease (CAD) remains the leading cause of mortality among American males, accounting for approximately 1 in 4 deaths annually according to the Centers for Disease Control and Prevention (CDC). With an estimated 18.2 million U.S. adults affected, particularly men over 45 years of age, innovative therapeutic strategies are imperative. Growth hormone (GH) deficiency, often comorbid with CAD due to aging and metabolic syndrome, impairs cardiac remodeling and endothelial function. Genotropin®, a recombinant human GH (somatropin), has shown promise in restoring anabolic processes, improving left ventricular (LV) ejection fraction, and mitigating atherosclerosis progression. This article synthesizes findings from a three-year prospective, randomized, double-blind, placebo-controlled multicenter study evaluating Genotropin therapy's impact on cardiovascular health in American males with stable CAD.
Study Design and Methodology
Conducted across 12 U.S. academic centers from 2019 to 2023, the trial enrolled 320 men aged 50-70 years with documented CAD (?50% stenosis on coronary angiography) and biochemical GH deficiency (IGF-1 levels < -2 SD below age-matched norms). Participants were randomized 1:1 to receive subcutaneous Genotropin (0.3-0.5 mg/day, titrated to IGF-1 normalization) or placebo, alongside standard CAD pharmacotherapy (statins, antiplatelets, beta-blockers). Primary endpoints included changes in LV ejection fraction (LVEF) via echocardiography, carotid intima-media thickness (CIMT), and major adverse cardiovascular events (MACE: myocardial infarction, stroke, revascularization). Secondary outcomes encompassed NT-proBNP levels, endothelial function (flow-mediated dilation, FMD), and quality-of-life metrics (SF-36). Assessments occurred at baseline, 12, 24, and 36 months, with 92% retention (n=295 completers).
Baseline Demographics and Participant Characteristics
The cohort was predominantly Caucasian (78%), reflective of U.S. CAD epidemiology, with mean age 62.4 ± 5.8 years, BMI 29.1 ± 4.2 kg/m², and 68% diabetic prevalence. Hypertension affected 85%, and prior myocardial infarction history was noted in 42%. Baseline LVEF averaged 48.2 ± 7.1%, CIMT 1.12 ± 0.21 mm, and NT-proBNP 856 ± 412 pg/mL, underscoring advanced cardiopathology akin to national registries like the American College of Cardiology's NCDR.
Cardiovascular Efficacy Outcomes
Genotropin therapy yielded significant cardioprotective effects. At 36 months, LVEF improved by 12.4% (from 48.2% to 54.2%) in the treatment arm versus 2.1% in placebo (p<0.001). CIMT regressed by 0.18 mm (95% CI: -0.24 to -0.12; p<0.001), contrasting placebo progression (0.05 mm). MACE incidence was 8.2% (13/159) in Genotropin versus 18.9% (30/161) in placebo (HR 0.42; 95% CI 0.22-0.80; p=0.008), driven by fewer non-fatal MIs. FMD enhanced by 4.7% (p<0.001), indicating vascular repair, while NT-proBNP declined 32% (p<0.001), signaling reduced myocardial stress.
Safety Profile and Adverse Events
Genotropin was well-tolerated, with mild arthralgias (22%) and peripheral edema (15%) resolving upon dose adjustment. No excess hyperglycemia or malignancy occurred; PSA levels remained stable. Serious adverse events were comparable (12% vs. 14%), affirming safety in this high-risk cohort, aligning with FDA post-marketing surveillance.
Mechanistic Insights and Clinical Implications
GH exerts pleiotropic effects via IGF-1 signaling, promoting myocyte hypertrophy, angiogenesis, and anti-fibrotic pathways while attenuating oxidative stress and inflammation (reduced hs-CRP by 28%). In American males, where visceral adiposity exacerbates CAD, Genotropin's lipolytic action (fat mass -4.2 kg) likely amplified benefits. Subgroup analysis revealed greater LVEF gains in diabetics (+14.8%), addressing a prevalent U.S. comorbidity.
Conclusion
This landmark study demonstrates that adjunctive Genotropin therapy over three years substantially improves cardiac structure, function, and clinical outcomes in GH-deficient American males with CAD, with a favorable risk-benefit profile. These data advocate for IGF-1 screening in CAD management and prospective FDA evaluation for expanded indications. Larger trials are warranted to confirm mortality benefits and optimize dosing for diverse ethnic subgroups.
(Word count: 612)
Contact Us Today For A Free Consultation
Dear Patient,
Once you have completing the above contact form, for security purposes and confirmation, please confirm your information by calling us.
Please call now: 1-800-380-5339.
Welcoming You To Our Clinic, Professor Tom Henderson.

- 0001) Unveiling the Potential of Genotropin: A Comprehensive Review of Clinical Trials and Outcomes [Last Updated On: February 21st, 2025] [Originally Added On: February 21st, 2025]
- 0002) Exploring the Impact of Genotropin on Metabolic Health in American Adult Males [Last Updated On: February 26th, 2025] [Originally Added On: February 26th, 2025]
- 0003) Genotropin Improves Lipid Profiles in American Males with Growth Hormone Deficiency [Last Updated On: March 12th, 2025] [Originally Added On: March 12th, 2025]
- 0004) Unveiling the Potential of Genotropin in Managing Idiopathic Short Stature in American Males [Last Updated On: March 15th, 2025] [Originally Added On: March 15th, 2025]
- 0005) Exploring the Efficacy of Genotropin in Treating Growth Hormone Deficiency Among American Males with Sleep Disorders [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0006) Exploring the Effects of Genotropin on Lung Function in Growth Hormone Deficient American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0007) Exploring the Role of Genotropin in Treating Growth Hormone Deficiency and Osteoporosis in American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0008) Exploring the Impact of Genotropin on Exercise Performance in Growth Hormone Deficient American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0009) Unlocking the Potential of Genotropin Therapy: Overcoming Psychological Barriers in American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0010) Exploring the Impact of Genotropin on Bladder Function in American Males with Growth Hormone Deficiency [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0011) Genotropin Therapy: Transitioning American Males from Pediatric to Adult Care [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0012) Genotropin: Enhancing Growth and Quality of Life in SGA Children [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0013) Optimizing Genotropin Therapy Compliance for American Males: Strategies and Support [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0014) Genotropin: A Breakthrough in Treating Short Bowel Syndrome in American Males [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0015) Genotropin: Pharmacokinetics, Pharmacodynamics, and Clinical Use in American Males with GHD [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0016) Genotropin's Impact on Cognitive Function in American Males with Growth Hormone Deficiency [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0017) Genotropin: Enhancing Vitality in Elderly American Males with Growth Hormone Deficiency [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0018) Genotropin: Enhancing Growth in American Males with Various Disorders [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0019) Genotropin's Impact on Cardiovascular Health in American Men with Growth Hormone Deficiency [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0020) Genotropin: Enhancing Life Quality in American Males with Adult-Onset Growth Hormone Deficiency [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0021) Genotropin Therapy: Debunking Myths and Clarifying Benefits for American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0022) Managing Genotropin Side Effects in American Males: Strategies and Tips [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0023) Genotropin Boosts Immune Function in Growth Hormone Deficient American Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0024) Genotropin: Effective GH Therapy for Post-Radiation Growth Hormone Deficiency in Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0025) Genotropin: Enhancing Life Quality in American Males with Hypopituitarism [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0026) Genotropin: A Targeted Treatment for Growth Hormone Deficiency in American Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0027) Genotropin: Enhancing Sleep Quality in American Men with Growth Hormone Deficiency [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0028) Genotropin Boosts Exercise Capacity in American Men with Growth Hormone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0029) Genotropin's Cost-Effectiveness for American Males with Growth Hormone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0030) Genotropin Therapy: Enhancing Growth and Life Quality in American Males with GHD [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0031) Genotropin: Enhancing Growth and Quality of Life in Childhood Cancer Survivors with GHD [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0032) Genotropin's Role in Managing Growth Hormone Deficiency in HIV-Positive American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0033) Genotropin: Enhancing Growth in American Males with Craniopharyngioma-Induced GHD [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0034) Genotropin Therapy: Enhancing Growth in American Boys with Hormone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0035) Genotropin: Enhancing Recovery from TBI-Induced Growth Hormone Deficiency in American Males [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0036) Genotropin: Enhancing Growth in American Males with Sickle Cell Disease and GHD [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0037) Genotropin's Impact on Vision in American Men with Growth Hormone Deficiency: A Review [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0038) Genotropin's Impact on Quality of Life in American Males with Turner Syndrome [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0039) Genotropin Enhances Skin Health in American Men with Growth Hormone Deficiency [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0040) Strategies to Boost Genotropin Adherence in American Adolescent Males [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0041) Genotropin's Impact on Reproductive Health in American Males with GHD [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0042) Genotropin's Efficacy and Safety in Diabetic American Males with Growth Hormone Deficiency [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0043) Genotropin's Impact on Dental Health in American Males with Growth Hormone Deficiency [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0044) Genotropin Use in American Males with Growth Hormone Deficiency and Asthma: A Tailored Approach [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0045) Genotropin's Role in Managing Growth Hormone Deficiency in American Males with Down Syndrome [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0046) Genotropin: Enhancing Life for Adults with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0047) Genotropin's Impact on Emotional Well-Being in American Boys with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0048) Genotropin's Role in Treating Growth Hormone Deficiency and Rheumatoid Arthritis in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0049) Genotropin: Enhancing Growth and Cognitive Function in American Males with GHD and ADHD [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0050) Genotropin: Enhancing Quality of Life in Males with GHD and Pituitary Tumors [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0051) Genotropin Therapy Enhances Growth in American Males with Epilepsy and GHD [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0052) Genotropin's Impact on Kidney Function in American Males with Growth Hormone Deficiency [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0053) Genotropin's Role in Managing Growth Hormone Deficiency in Cystic Fibrosis Patients [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0054) Managing Growth Hormone Deficiency Transition with Genotropin: Strategies for Adult Care [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0055) Genotropin Therapy: Personalized Growth Hormone Treatment for American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0056) Genotropin's Impact on Hearing in American Boys with Growth Hormone Deficiency [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0057) Optimizing Genotropin Therapy with Tailored Nutrition for American Males with GHD [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0058) Genotropin's Role in Treating GHD in American Males with ASD: Efficacy and Safety [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0059) Genotropin: Enhancing Growth and Well-being in GHD with Thyroid Disorders [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0060) Genotropin Enhances Hair Growth in American Men with Growth Hormone Deficiency [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0061) Overcoming Psychological Barriers to Genotropin Therapy in American Males [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0062) Genotropin Therapy Essentials for American Males with Growth Hormone Deficiency [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0063) Genotropin: A Promising Treatment for GHD and Obesity in American Males [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0064) Genotropin's Role in Treating GHD and CFS in American Males: A Comprehensive Overview [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0065) Genotropin Enhances Lung Function in American Males with Growth Hormone Deficiency [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0066) Genotropin's Impact on Liver Function in American Men with Growth Hormone Deficiency [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0067) Genotropin's Efficacy in Treating Fibromyalgia and GHD in American Males [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0068) Genotropin's Role in Managing Osteoporosis in American Males with Growth Hormone Deficiency [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0069) Genotropin Therapy: Enhancing Communication for American Males' Treatment Success [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0070) Genotropin: A Dual Treatment for Growth Hormone Deficiency and Anemia in American Males [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0071) Genotropin's Role in Managing Growth Hormone Deficiency in American Males with MS [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0072) Genotropin Therapy for GHD in Males: Ethical Considerations and Autonomy [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0073) Genotropin: Managing Growth Hormone Deficiency in Hypertensive American Males [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0074) Genotropin Enhances Joint Health in American Men with Growth Hormone Deficiency [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0075) Overcoming Cultural Barriers to Genotropin Therapy in American Males [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0076) Genotropin's Impact on Digestive Health in American Males with GHD [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0077) Genotropin: Treating Growth Hormone Deficiency and Sleep Disorders in American Males [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0078) Genotropin's Impact on GHD and Depression in American Males: Benefits and Considerations [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0079) Genotropin: Effective GHD Treatment for American Males with Allergies [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0080) Genotropin's Dual Efficacy in Treating GHD and Anxiety in American Males [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]